All submitted manuscripts must strictly adhere to the core practices outlined by the COPE, which serves as a comprehensive guide for Scifiniti in upholding the highest standards of publication ethics and integrity in scholarly publishing. This encompasses ethical practices in publishing, peer review integrity, editorial board appointments, conflicts of interest, authorship and contributorship disputes, conflicts of interest, handling retractions, and addressing allegations of publication misconduct, including data falsification, fabrication, and plagiarism. Publication misconduct is strictly prohibited and stringent measures will be taken against individuals engaged in unethical activities.
1. Authorship
Scifiniti abides by the guidelines recommended by the ICMJE and COPE for authorship criteria. To become an author, an individual must follow the four criteria stated by ICMJE:
- Substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work; AND
- Drafting the work or reviewing it critically for important intellectual content; AND
- Final approval of the version to be published; AND
- Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The corresponding author is a primary author who coordinates with the Editorial office during manuscript processing from submission to publication process. They act as a liaison between the co-authors and the journal, keeping them updated with the manuscript status and decisions.
A coauthor is an individual who makes substantial contributions to the conception, design, investigation, methodology, execution, or analysis of research presented in the manuscript.
In the case of those manuscripts, where authorship is retained by the consortium or group, then the author list must contain the name of the consortium or group if individual authors meet ICMJE’s criteria of authorship (as described above).
It is recommended that individuals who do not meet all the specified criteria for authorship should not be included as authors in the paper. Instead, their contributions should be acknowledged in the acknowledgement section of the manuscript. This practice is in line with the recommendations from organizations such as the ICMJE and the COPE. Before proceeding with acknowledgement, it is essential to ensure the approval of the corresponding author and to adhere to the specific authorship and acknowledgement policies of the target journal to maintain transparency and ethical standards in scholarly publishing.
The following activities alone, without additional substantial contribution, don’t meet authorship criteria:
-
- Acquisition of Funding
- Supervision of a Research Group
- Administrative Support
- Language Editing
- Proofreading
- Writing Assistance
- Technical Editing
For further details, please review ICMJE's definition of non-author contribution.
If an author is deceased during the manuscript processing stage, then it is the responsibility of the corresponding author or co-authors to notify the editorial office. In cases where the deceased author was the corresponding author, the remaining authors, with mutual consent, should nominate another author to assume the role of the corresponding author. This information should be provided and appended, including the name of the newly nominated corresponding author, following the publication of the manuscript.
-
Gift, Guest, and Ghost Authorship
Gift authorship, also known as honorary authorship involves individuals who have made no significant contribution to the preparation of the manuscript. There could be several reasons to include a guest author such as establishing credibility or satisfying institutional or publication requirements.
Similarly, Guest authorship is an authorship with no contribution in manuscript preparation but their names are added for courtesy, gratitude, or other non-substantive reasons.
In contrast to gift and guest authors, ghost authors are those who have made significant contributions to the manuscript's preparation but their names are not listed in authorship. Instead, their contribution has been attributed to someone else in the manuscript.
Identifying guest, gift, and ghost authors is sometimes difficult. Scifiniti therefore follows COPE Guidelines to identify such authorship problems. If such authorship issues are identified, appropriate actions as recommended by COPE will be taken.
-
Artificial Intelligence (AI)-Assisted Technology
Scifiniti strongly adheres to COPE statement regarding the use of artificial intelligence in the manuscripts. AI tools such as ChatGPT and other large language models (LLMs) such as BERT, GPT-3, and T5 cannot take responsibility for submitted work, identify conflicts of interest, or manage copyright and licensing agreements so these tools can’t be listed as authors.
Recommendations
- The use of AI tools should be disclosed in the cover letter.
- Specify the use of the Al tool in the Acknowledgement section.
- The use of AI tools must be mentioned in the “Materials and Methods” section.
Authors are completely responsible for the transparency, originality, integrity, and validity of their manuscript's content, including sections generated by AI tools, and are consequently responsible for any violations of publication ethics.
Authorship disputes, if they occur, are addressed in accordance with the guidelines recommended by the COPE. Generally, disputed authorship does not justify retraction as long as there is no valid reason to question the validity of the findings.
2. Ethical Guidelines for Authors
-
Redundant and Duplicate Publication
Submitting the same manuscript in two or more journals simultaneously is strictly prohibited and will be considered an unethical publishing practice. Authors are therefore required to confirm that the manuscript they submit has not been submitted, in process, or previously published in any other journal.
During manuscript submission, authors are required to ascertain that it is an original contribution of the author/authors. If any portion is reproduced or reused from a previously published article, proper attribution must have been provided, has been properly attributed and permission from the original copyright holder, if necessary, must have been obtained.
Authors’ contributions toward the preparation of manuscripts must be properly mentioned in the manuscript. Authors are recommended to refer to CRediT (Contributor Roles Taxonomy), which offers a comprehensive taxonomy encompassing 14 distinct roles that characterize contributors' involvement in research outputs. These roles serve to specify the contributions made by each author to the article.
How to Specify Authors’ Contribution?
- X.X. contributed to Conceptualization and Writing.
- X.Y. conducted Data curation and Analysis.
- Y.Y. has a contribution in Investigation and Methodology.
-
Permission for Reproduction
Authors are recommended to get signed, written permission (if necessary) from the copyright holder for the reproduction of already published figures, tables, or schemes.
Authors are highly recommended to provide appropriate attribution by citing others' work that has significantly impacted their manuscript.
Authors must disclose clearly if they have any possible conflicts of interest related to their manuscript at the time of manuscript submission.
Self-plagiarism is an uncredited use of authors’ own published work without proper citation in the manuscript. It is therefore recommended that the author properly cite his published work if reproduced or reused in the manuscript.
Authors are required to mention appropriately the name of the ethical committee that they have used for researching humans, animals, and plants. The name of the committee from where approval was taken and the reference number should be mentioned in the manuscript. Authors are also required to get consent from patients or legal guardians in case they use details, images, or videos of a person.
3. Change in the Authors' List
Any change in the authors’ list such as a change in sequence, addition, or deletion during or after the publication of the manuscript, must be approved by the listed authors. The corresponding author is therefore suggested to provide:
- Written consent of all authors, including those who are either added or deleted.
- A revised copyright letter with the updated list of authors.
- Authors’ contribution statement if added, or reason for removal.
Scifiniti however, reserves the right to accept or reject modification based on provided evidence and in this connection, COPE recommended protocol will be used:
For comprehensive information on Authorship, please review “COPE Discussion Document on Authorship”.
4. Data Fabrication and Falsification
Data fabrication and falsification are unethical practices undermining Scifiniti’s core scientific integrity principles. Data fabrication involves the deliberate creation of entirely new data detached from legitimate experimental or observational processes. Data falsification entails manipulating or altering existing data to misrepresent findings such as image manipulation, removing outliers or manipulated results, changing data points, etc. Both actions compromise the accuracy of research and can have severe consequences.
Authors are strongly encouraged to ensure that there is no intentional data fabrication and data falsification in their work.
In case data Fabrication or falsification is detected, it will be dealt with per the COPE recommendation, which may result in the rejection or retraction of manuscripts. Therefore, authors are strongly advised to exercise utmost care while submitting manuscripts.
COPE Recommendations
Fabricated Data in a Submitted Manuscript
Fabricated Data in a Published Manuscript
5. Plagiarism
Plagiarism, the act of using someone else's work without proper attribution, is strictly prohibited within Scifiniti's publishing guidelines. Authors are strongly advised to submit only original manuscripts, ensuring that their work is a genuine contribution to the academic discourse.
Scifiniti employs professional plagiarism detection software to uphold the integrity of the peer review process. This software meticulously examines submitted manuscripts for any instances of plagiarism. In the event that plagiarism is identified during the peer review process, the manuscript will be subject to further evaluation. The decision of whether to reject the manuscript outright or request revisions will be made after a comprehensive content assessment, going beyond a mere percentage review.
It is essential to note that if plagiarism is detected after publication, it may result in the retraction of the manuscript. Authors are, therefore, strongly urged to diligently check their manuscripts using any reputable plagiarism detection tool before submission. This precautionary measure aims to prevent rejection due to similarities in content and ensures the scholarly integrity of the publications associated with Scifiniti.
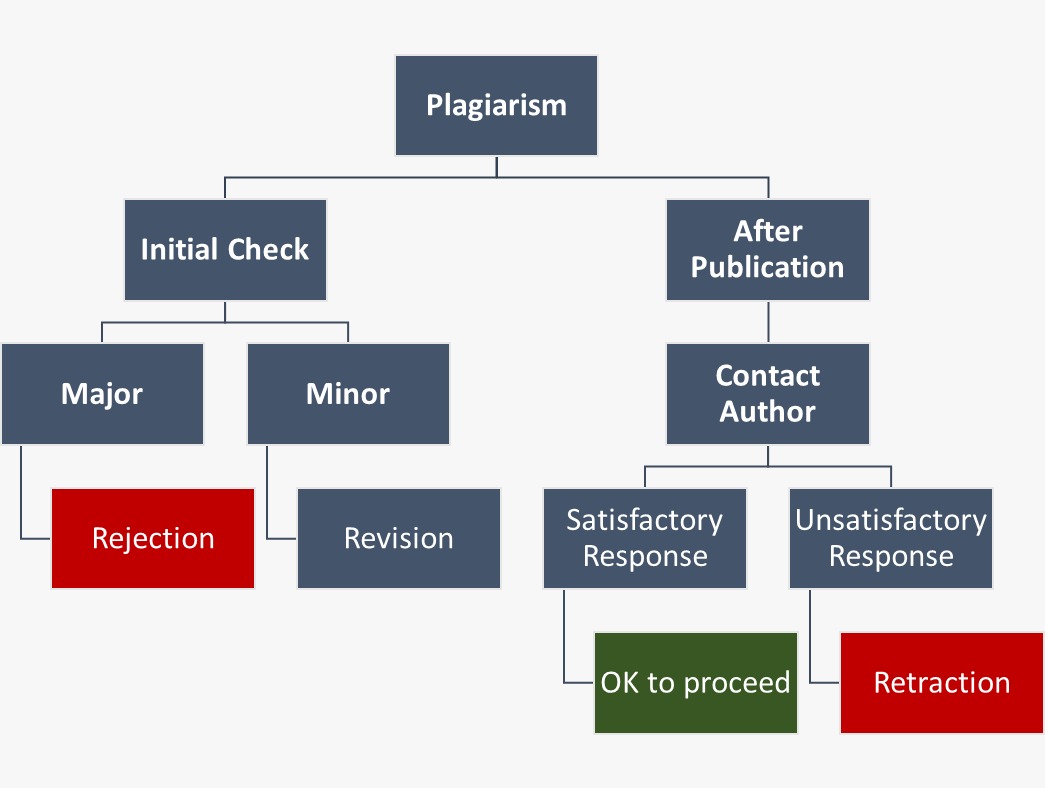
Scifiniti strictly follows COPE guidelines to deal with cases of plagiarism.
Similarly, image files must remain unaltered or modified in such a way that could not change the information of the original image. These inappropriate modifications include modifying any of the features of the image, combining it with any other image, enhancing brightness, contrast, colour balance, etc. If image manipulation is found during the peer review process, then the manuscript will either be rejected or modified. If image manipulation is found after publication, then it will be handled, as per the suggested protocol of COPE.
- How to deal with Image manipulation in a published article [View Flowchart]
The following are the useful links on image manipulation:
6. Ethical Policies
When documenting research that involves human subjects, human materials, human tissues, or human data, it is imperative for authors to state that the research adhered to the principles outlined in the Declaration of Helsinki of 1975 (revised in 2013). More information about the Declaration of Helsinki can be found at https://www.wma.net/what-we-do/medical-ethics/declaration-of-helsinki/.
According to point 23 of this declaration, “The research protocol must be submitted for consideration, comment, guidance, and approval to the concerned research ethics committee before the study begins. This committee must be transparent in its functioning, must be independent of the researcher, the sponsor, and any other undue influence, and must be duly qualified. It must take into consideration the laws and regulations of the country or countries in which the research is to be performed as well as applicable international norms and standards but these must not be allowed to reduce or eliminate any of the protections for research subjects set forth in this Declaration.
The committee must have the right to monitor ongoing studies. The researcher must provide monitoring information to the committee, especially information about any serious adverse events. No amendment to the protocol may be made without consideration and approval by the committee. After the end of the study, the researchers must submit a final report to the committee containing a summary of the study’s findings and conclusions.”
Authors are recommended to provide a separate section for Human rights and mention clearly that research has been conducted by the Declaration of Helsinki, along with the identification code and name of the Ethics Committee that has approved the protocol.
-
Research Involving Animals
We expect authors should follow the Animal Use Alternatives (3Rs) model while conducting any research that requires the use of animals. These new scientific approaches proposed by Drs. William Russell and Rex Burch, help to minimize to maintain scientific integrity by reducing the pain and distress of research animals.
We recommend authors follow the “Basel Declaration” which summarizes essential principles to follow when conducting research in animals. The International Council for Laboratory Animal Science (ICLAS) has also published ethical guidelines that could help authors protect animal rights while conducting any research.
Authors are required to explicitly mention the name of the approval committee and reference number in their manuscript, emphasizing that they obtained both legal and ethical approval before commencing the research involving animals. Additionally, the authors must affirm that the experiments were conducted in strict adherence to the guidelines outlined below.
If ethical approval is exempted from any committee then this should also be clearly mentioned in the manuscripts along with the name of the committee that grants exemption.
Scifiniti recommends authors use ARRIVE guidelines for reporting research involving animals. Reviewers are also recommended to use CHECKLIST while reviewing a manuscript and provide their recommendations if the research is not in compliance with any of the points mentioned in the checklist.
For in vivo experiments, visit https://www.nc3rs.org.uk/arrive-guidelines
-
Research Involving Plants
Experimental research on plants must be conducted in compliance with relevant guidelines published by the IUCN Policy Statement on Research Involving Species at Risk of Extinction, the Convention on Biological Diversity, and the Convention on the Trade in Endangered Species of Wild Fauna and Flora.
Authors are advised to provide ethical statements including the name and source of the plant used during research.
Example: Solanum tubersum was used in conducting research which was provided by Dr. (XYZ), affiliation, City, Country.
To support reproducibility, voucher specimens of rare and non-model plants must be deposited in a publicly accessible herbarium, public museum or another public repository. These vouchers should include documentation of specific parts of the plants which are used in the research, details of the geographical coordinates of the collection site and the collection date.
-
Research Involving Cell Lines
If a research involves the use of cell lines then authors should mention the origin in the methods sections of their manuscript. If an author has used established cell lines then the source and reference of the published article should be mentioned. For previously unpublished de novo cell lines, details of the ethical committee and approval should be mentioned in the manuscript. Informed consent must be taken in cases where human cell lines are used.
Example: The HaCaT cell lines were provided by XYZ
For misidentification and contamination of human cell lines, please International Cell Line Authentication Committee (ICLAC) and NCBI database.
For use, ISSCR Guidelines for Stem Cell Research and Clinical Translation should be followed for reporting experiments on human embryos, gametes and embryonic stem cells.
7. Consent for Publication
If authors have used details, images, or videos of a person, then written consent from the relevant individual must also be taken to publish under Creative Commons Attribution License 4.0. If an individual is under 18 then parents or legal guardians must be contacted for their consent to publish data. Personal identifiers, such as names, initials, or hospital numbers, should not be disclosed in the manuscript unless it is indispensable for scientific purposes.
In cases where an individual has passed away, it is necessary to seek consent for publication from their immediate family or next of kin. The manuscript should incorporate a declaration confirming that written informed consent for publication has been secured.
For unidentified images like X-rays, ultrasound images, etc., no consent for publication is needed.
For further details, ICMJE recommendations for “Protection of Research Participants”.
8. Trial Registration
Scifiniti strongly recommends its authors follow ICMJE's recommendations for registration of clinical trials in a public trials registry. They are also recommended to cite it as a reference in their manuscript.
According to WHO “a clinical trial is any research study that prospectively assigns human participants or groups of humans to one or more health-related interventions to evaluate the effects on health outcomes. Clinical trials may also be referred to as interventional trials. Interventions include but are not restricted to drugs, cells and other biological products, surgical procedures, radiologic procedures, devices, behavioural treatments, process-of-care changes, preventive care, etc.” This definition includes Phase I to Phase IV trials."
In line with the position of ICMJE, Scifiniti strongly encourages authors to include a statement in their manuscript that the results are unpublished in a peer-reviewed journal; this information can however be updated as results are published.
Scifiniti recommends CONSORT for randomised controlled trials; authors submit CONSORT checklist and Protocol Flow Diagram during manuscript submission.
-
Sex and Gender in Research (SAGER)
Authors are advised to follow ‘Sex and Gender Equity in Research – SAGER – guidelines’. Authors should ensure that the terms sex (referring to a biological attribute) and gender (influenced by social and cultural factors) are used carefully in order to avoid confusion in the two terms. In the introduction, authors should consider discussing the potential presence of sex and/or gender differences. Furthermore, they should detail the methods used to address sex and/or gender in the study's design and present data separated by sex and/or gender when it is relevant. Authors should also analyze and present results specific to each sex and/or gender group. If the study did not include a sex and/or gender analysis, the authors should provide a justification in the discussion. We recommend that our authors refer to the comprehensive guidelines before submitting their work.
9. Funding
Authors are required to disclose all sources of funding in the acknowledgment section of the manuscript. This includes grants, scholarships, sponsorships, or any financial contributions. Details such as the name of the funding agency, grant number, and specific requirements set by the funding body should be clearly stated.
10. Conflicts of Interest
As a scientific publisher, we maintain a strong commitment to upholding the highest standards of integrity, objectivity, and transparency in the dissemination of scientific knowledge. We recognize the importance of managing potential conflicts of interest to safeguard the integrity of the research and scholarly content we publish. Authors are strongly recommended to disclose any potential conflict of interest in their manuscripts under the section “Conflicts of Interest”.
Examples of Financial Conflicts of Interest
-
- Employment or Voluntary Engagement.
- Partnerships with Advocacy Organizations on Article Content.
- Research Grants from Funding Entities (author or organization recipients).
- Compensation, including Honoraria, Royalties, Consulting Fees, Lecture Fees, or Testimonies received by authors.
- Patents, including pending applications, held by authors, their institutions, funding bodies, or licensed to any entity, with or without royalty earnings.
- Royalty Income for authors or their affiliated institutions.
- Travel allowances
- Ownership of Stocks or Shares.
- Paid Lectures
Examples of Non-Financial Conflicts of Interest
-
- Personal, Political, Religious, Ideological, Academic, or Intellectual Conflicts of Interest.
- Engagement in Legal Proceedings related to the research or its outcomes.
- Academic competition.
Undisclosed Conflicts of Interest
Scifiniti follows COPE’s recommendation to deal with undisclosed conflict of interest cases before or after the publication.
Undisclosed conflict of interest in a submitted article (View COPE guidelines).
Undisclosed conflict of interest in a published article (View COPE guidelines).
11. Citation Policies
Authors are expected to follow the COPE Citation Policy while citing any article in their manuscripts. Citations manipulation by authors, reviewers and editors is strictly prohibited.
Authors should avoid artificially inflating citations by including an excessive number of references to their own work or to the work of colleagues and friends. However, citing previously published work is acceptable when it is genuinely relevant. In such cases, proper citation is essential to prevent self-plagiarism, redundant publication, or misrepresentation of scholarship. Scifiniti emphasizes that reviewers must carefully assess the relevance and appropriateness of all references and that the Editor-in-Chief must ensure citation practices adhere to ethical standards. Following these principles helps maintain the integrity, transparency, and credibility of published research.